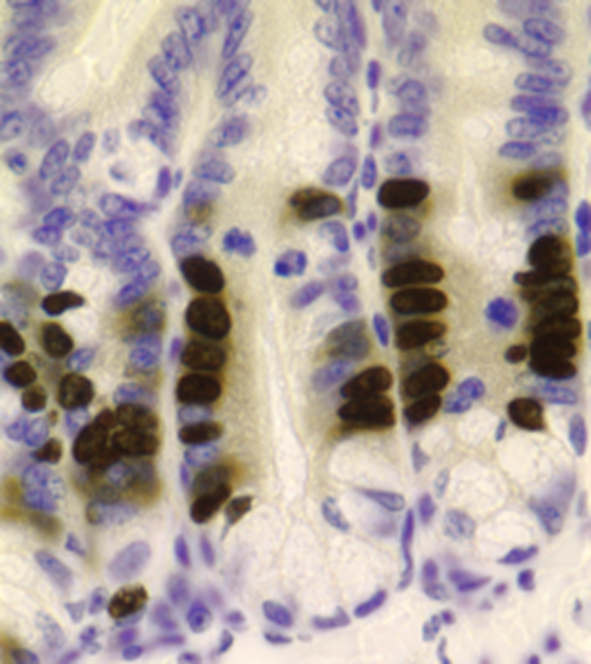
Alopecia Model
A defined area receives a high dose of radiation to induce alopecia. Novel therapeutic agents can be administered topically or systemically. Epistem can determine the effect of a novel therapeutic agent on the different stages of the hair cycle and the propensity to inhibit hair growth or cause hair loss.
Alternatively, hair can be maintained ex vivo and effects on cell proliferation, DNA damage etc examined, alongside observed modulation of target pathways.
Services include:
Mucositis Models
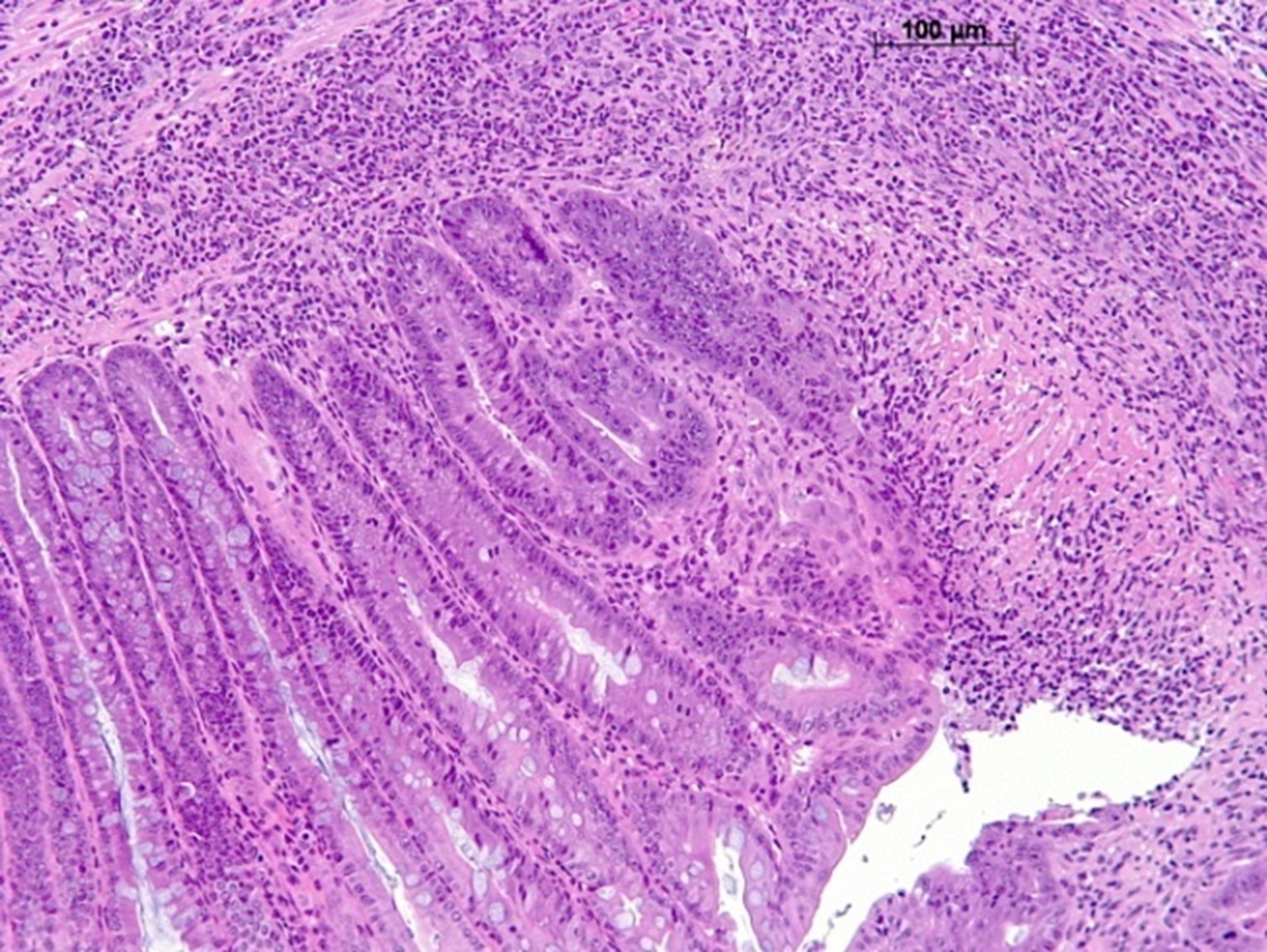
Our mucositis models are designed to replicate the painful and debilitating inflammation of the mucous membranes that can occur as a side effect of cancer treatments such as chemotherapy and radiation therapy. With this model, researchers can investigate potential treatments for mucositis and improve the quality of life for cancer patients undergoing these treatments. Epistem provides in vivo oral and gastrointestinal models which can be used to assess novel anti-mucositis therapies and to evaluate the clinical risk of mucositis induction by novel therapeutic agents.
Bone Marrow Toxicity
We can assess the extent and duration of myelosuppression, commonly referred to as bone marrow suppression, within our cancer models.
Effects on the Microbiome
Changes to the microbiome induced by oncological therapies can be monitored and potential treatments or dosing schedules examined that may minimise adverse perturbations.